Atriplex dioica
Amer. Monthly Mag. & Crit. Rev. 2(2): 119. 1817.
Herbs, monoecious, typically erect and often branching, 3–15 dm; stems green or striped, angular. Leaves opposite or subopposite at least proximally; petiole 1–3 cm; blade strongly 3-veined from near base, lanceolate to linear-lanceolate or often triangular-ovate or lance-ovate, 30–125 × 25–60(–80) mm, thickened, basal or subbasal lobes spreading to mainly antrorse, obtuse, blade otherwise entire to sparingly dentate, typically scurfy. Flowers in terminal or terminal on lateral branches, in spiciform spikes, 2–9 cm, naked except at base. Fruiting bracteoles green, blackening in age, sessile, veined or veins obscure, broadly triangular to ovate, 3–10 mm, thick, usually longer than wide, base truncate to obtuse, margin united at base, apex acute lateral angles entire or with short, sharp teeth, faces smooth or with 2 tubercles, with an inflated inner spongy layer. Seeds ellipsoid, wider than long, dimorphic: brown, 1.5–3 mm wide, flattened at base, or black, 1–2 mm wide, shiny; radicle inferior. 2n = 36, 54.
Phenology: Flowering summer–fall.
Habitat: Sea beaches, other saline habitats
Elevation: 0-1500 m
Distribution
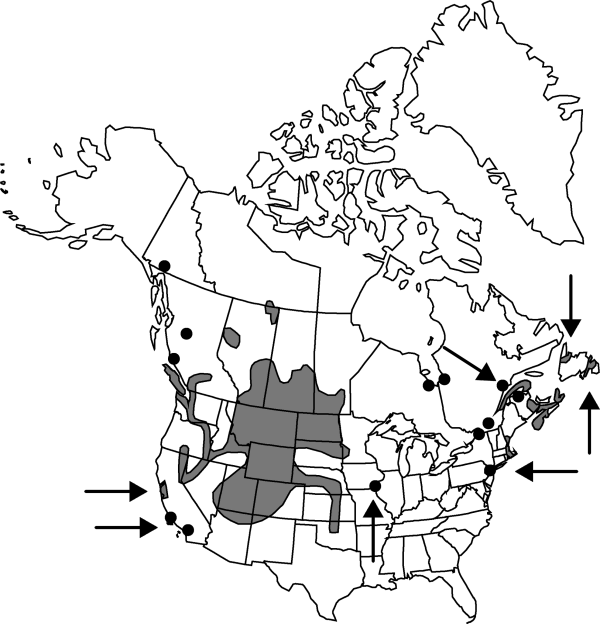
Alta., B.C., Man., Nfld. and Labr. (Nfld.), N.S., Nunavut, Ont., Que., Sask., Yukon, Ariz., Calif., Colo., Idaho, Iowa, Kans., Maine, Mass., Minn., Mont., Nebr., Nev., N.H., N.J., N.Mex., N.Dak., Okla., Oreg., R.I., S.Dak., Utah, Wash., Wyo.
Discussion
The distribution of the species is evidently bipartite, with an eastern coastal series extending northward mainly from New Jersey to Newfoundland and along the St. Lawrence seaway, and perhaps extending to James Bay of Hudson Bay. The western grouping lies mainly west of the 95th meridian of longitude, where it has been collected since early historical times to the present in saline marshes or other saline sites from the Yukon Territory and Northwest Territories, southward to southern California, northern Arizona, northern New Mexico, and Oklahoma.
Rafinesque gave the following information: “Stem upright angular branched, leaves petiolate, deltoid, acute, thick, scaly, the proximal opposite toothed, the distal alternate, hastated, entire: flowers dioical glomerate, male spiked naked, female unequal, sepals deltoid, warty-crested.”
The name Atriplex dioica Rafinesque antedates Chenopodium subspicatum Nuttall by half a year, being published in December 1817. Hence, it is the correct name for the widely ranging species, which has passed most recently under the name A. subspicata. Nuttall’s description of the habitat of Chenopodium subspicatum is: “In saline soils around Mandan Village, Missouri,” a designation of habitat that applies to this day.
The species forms a mirror-image set of specimens with the remarkably similar Atriplex prostrata, from which it may be distinguished in most cases by the thickened, merely ovate to lanceolate leaf blades, and less commonly but in some localities exclusively triangular-hastate to lanceolate, mostly scurfy and prominently 3-veined leaf blades. In some specimens, including the types of both Chenopodium subspicatum and Atriplex carnosa, the blades bear a hastate lobe at or above the base and sometimes match triangular-hastate profile of A. prostrata. The leaves of A. prostrata are typically thin-textured, green, not scurfy, and the veins of the blade are obscure. I. J. Bassett et al. (1983) disallowed within A. dioica (as A. subspicata) any but those with lanceolate blades, including those with the proximalmost leaves with a pair of subbasal hastate lobes. However, there are numerous specimens in which the blades are thickened and transitional in that regard to the triangular-hastate profile as in A. prostrata. Certainly those specimens with triangular or triangular-hastate leaves taken prior to the introduction of A. prostrata sometime late in the nineteenth or early twentieth century, clearly belong to the indigenous A. dioica. Whether there are intermediates between diploid (2n = 18) A. prostrata and tetraploid or hexaploid (2n = 36, 54) A. dioica is not known. There does not seem to be any consistent feature or combination of features by which all specimens can be assigned to one or the other of the two taxa. It seems probable, however, that A. prostrata is a late introduction from Europe, and that it, along with the related A. heterosperma, is now invading habitats previously occupied exclusively by the indigenous A. dioica.
Selected References
None.
