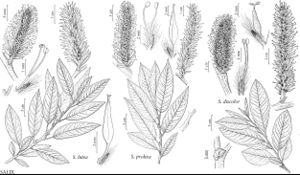
Salix discolor
Ges. Naturf. Freunde Berlin Neue Schriften 4: 236, plate 6, fig. 1. 1803.
Shrubs, 2–4(–8) m, (sometimes forming clones by stem fragmentation). Stems: branches dark red-brown or yellow-brown, not to strongly glaucous, villous to glabrescent, (peeled wood smooth or striate, striae sometimes very dense, to 10 mm); branchlets yellowish, red-brown, or yellow-brown, or dark brown, moderately densely velvety, velutinous, or tomentose to glabrescent. Leaves: stipules rudimentary on early ones, foliaceous on late ones, (0.8–12.5 mm), apex acute to acuminate; petiole convex to flat adaxially, 6–17 mm, tomentose adaxially; largest medial blade narrowly elliptic, elliptic, oblanceolate, or obovate, 30–80(–135) × 12–33 mm, (2.3–)3–3.5(–4.5) times as long as wide, base convex or cuneate, margins flat, crenate, irregularly toothed, sinuate, or entire, apex acute, convex, or acuminate, abaxial surface glaucous, glabrous, pilose, sparsely pubescent or long-silky, midrib glabrous or densely pubescent, hairs (white, sometimes also ferruginous), wavy, adaxial dull or slightly glossy, glabrous or pilose, (hairs rarely ferruginous); proximal blade margins entire or serrulate; juvenile blade reddish or yellowish green, pilose, tomentose or moderately densely short-silky abaxially, hairs white and ferruginous. Catkins flowering before leaves emerge; staminate stout or subglobose, 23–52 × 12–22 mm, flowering branchlet 0–3 mm; pistillate densely flowered (loose in fruit), slender or stout, 25–108(–135 in fruit) × 12–33 mm, flowering branchlet 0–10 mm; floral bract brown, black, or bicolor, 1.4–2.5 mm, apex acute or convex, abaxially hairy, hairs straight. Staminate flowers: adaxial nectary oblong, 0.6–1.1 mm; filaments distinct, glabrous or hairy basally; anthers yellow or purple turning yellow, ellipsoid or short- or long-cylindrical, 0.5–1 mm. Pistillate flowers: adaxial nectary oblong or ovate, 0.7–1.3 mm, shorter than stipe; stipe 1.6–2.7 mm; ovary obclavate or pyriform, short-silky (hairs straight), beak sometimes slightly bulged below styles; ovules 6–16 per ovary; styles 0.3–1 mm; stigmas slenderly or broadly cylindrical, 0.48–0.64–0.88 mm. Capsules 6–11 mm. 2n = 76, 95, 114.
Phenology: Flowering early Apr-late May.
Habitat: Marshy margins of ponds, streams, and open alluvial woods, fens, seepage areas, peaty substrates
Elevation: 0-2400 m
Distribution
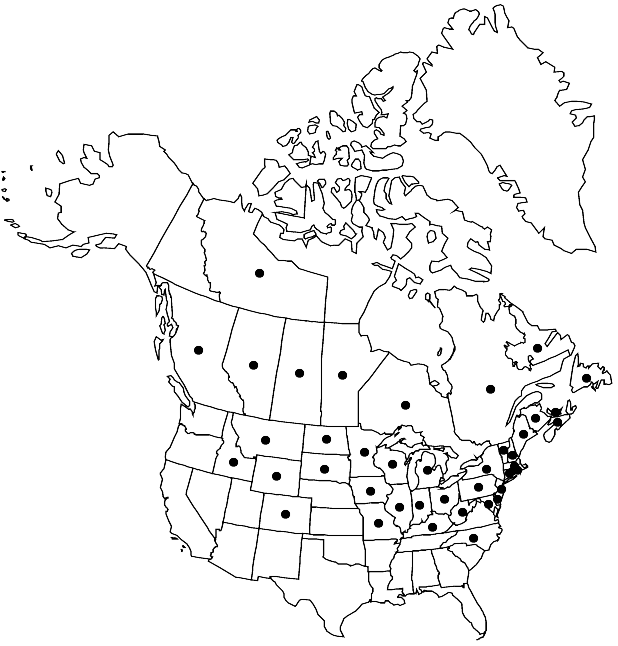
Alta., B.C., Man., N.B., Nfld. and Labr., N.W.T., N.S., Ont., P.E.I., Que., Sask., Colo., Conn., Del., Idaho, Ill., Ind., Iowa, Ky., Maine, Md., Mass., Mich., Minn., Mo., Mont., N.H., N.J., N.Y., N.C., N.Dak., Ohio, Pa., R.I., S.Dak., Vt., W.Va., Wis., Wyo.
Discussion
Salix discolor is introduced in North Carolina.
Vegetative specimens of Salix discolor can be difficult to distinguish from S. planifolia, but there are two, somewhat variable, characters that can be used. Salix discolor usually has leaves dull adaxially, with arcuate secondary veins widely and irregularly spaced; S. planifolia has leaves slightly or highly glossy adaxially, with straight secondary veins closely and regularly spaced.
Salix discolor in northeastern United States can be difficult to distinguish from widely naturalized S. atrocinerea and S. cinerea. Useful diagnostic characters are: tertiary leaf veins, which are irregular in S. discolor but close and parallel in introduced species, and raised striae on peeled 3–5-year old branches, which are absent or indistinct and relatively short in S. discolor, but long and very prominent in the introductions.
Hybrids:
Salix discolor forms natural hybrids with S. humilis, S. interior, S. myricoides, S. pellita, and S. planifolia. Reports of hybrids with S. candida and S. eriocephala (M. L. Fernald 1950), and S. bebbiana and S. pyrifolia (C. K. Schneider 1921; Fernald), are not based on convincing specimens. Synthetic hybrids with S. bebbiana could not be made (G. W. Argus 1974; A. Mosseler 1990) and those made with S. eriocephala had low seed viability (Mosseler).
Salix discolor × S. humilis has tomentose leaves of S. humilis and longer catkins and styles of S. discolor (G. W. Argus 1986). These species readily hybridize and produce abundant seed (Argus 1974). The hybrids are fertile and backcross. Specimens of S. discolor with densely villous branchlets may be hybrids or introgressants with S. humilis. The two species usually are ecologically isolated; S. discolor occurs in wetland thickets and S. humilis in dry, sandy upland forests. Where the two habitats come into proximity, hybrids occur but large swarms have not been observed.
Salix discolor × S. myricoides (S. ×laurentiana Fernald, syn. S. paraleuca Fernald) usually resembles S. myricoides but has hairy ovaries (R. D. Dorn 1975, 1976). This hybrid was originally described as a species, from lower St. Lawrence River, Quebec. Its most distinctive feature is that hairs appear on ovaries in patches, at the base or, sometimes, only on the stipes. A similar ovary indumentum pattern appears in other hybrids or species of hybrid origin, e.g., S. hookeriana. Characteristics of S. discolor found in S. ×laurentiana include epidermis with gray-margined splits, leaf margins entire or sinuate, leaves with 2–4 teeth per cm, anthers yellow or purple, filaments hairy on proximal half or basally, ovaries hairy, greenish brown or green with red sutures, and adaxial pistillate nectaries ovate. Characteristics of S. myricoides include inner bud-scale membranes separating from the outer ones, stipules more prominent, catkins on distinct flowering branchlets, and longer styles sometimes distinct about half their lengths. This hybrid occurs throughout the area of overlap of the parents. All three taxa often are intermixed but few hybrids seem to produce well-developed seed.
Salix discolor × S. pellita (S. ×pedunculata Fernald) is characterized by juvenile leaves with infolded or sometimes revolute margins, ovaries with patches of hairs relatively short, flattened, crinkled, and refractive, and catkins borne on distinct flowering branchlets 2–10 mm. This sporadic hybrid does not seem to be fertile. It occurs in Newfoundland, Quebec, and Saskatchewan. Although it has been collected at few localities, it probably is more common and should be expected wherever the two species grow together. The type and other collections compare very well with synthetic hybrids (A. Mosseler 1990), which were reported to show a high hybridization success rate, high F1 pollen viability, and high seedling viability. It was suggested that variability within these species may be due to interspecific gene flow. In interpreting the parentage of the wild hybrids it is not possible to rule out hybridization of S. planifolia or S. myricoides with S. pellita, or that these hybrids may be S. myricoides × S. planifolia, as suggested by B. G. O. Floderus (1939). Salix ×pellicolor Lepage is a later synonym of this hybrid.
Selected References
None.