Saxifragaceae
Herbs, perennial, rarely biennial (Saxifraga) or annual (Cascadia, Saxifraga), rhizomatous or not, stoloniferous or not, persistent stem ± erect as caudex, horizontal as rhizome, or intergrading, branched or unbranched, sometimes bearing bulbils (Bolandra, Lithophragma, Micranthes, Saxifraga, Suksdorfia). Flowering stems appearing in spring, summer, or autumn with leaves usually present (usually appearing in autumn or winter after basal leaves have withered in Jepsonia), leafless, or leafy and bearing 1–5 cauline leaves proximally, glabrous or short to long stipitate-glandular or eglandular, hairs usually multicellular (unicellular in Astilbe, Saxifragopsis). Leaves usually in basal rosettes, sometimes cauline, usually alternate, sometimes opposite (Chrysosplenium, Lithophragma, Mitella, Saxifraga), usually simple (compound in Astilbe, sometimes compound in Lithophragma, Tiarella); stipules absent or present; petiole absent or present, usually not jointed distally at attachment to blade (jointed distally in Saxifragopsis), usually not peltate (peltate in Darmera), not producing adventitious buds at apices of petioles of basal rosette and cauline leaves (usually producing adventitious buds at apices of petioles in Tolmiea); blade margins entire, crenate, serrate, or dentate, ciliate or glandular-ciliate. Inflorescences usually terminal racemes, panicles, cymes (simple or compound), thyrses (with lateral dichasial or monochasial cymose branches), or solitary flowers (Chrysosplenium, Lithophragma), sometimes axillary cymes (Chrysosplenium), usually arising from terminal or axillary buds in rosettes, 2–300(–1000+)-flowered, bracteate or ebracteate. Flowers usually bisexual, sometimes unisexual (Astilbe, Saxifraga), homostylous (heterostylous in Jepsonia), usually radially symmetric, sometimes bilaterally symmetric (Bensoniella, Heuchera, Micranthes, [Saxifraga], Tiarella, Tolmiea); perianth and androecium hypogynous, perigynous, or epigynous; hypanthium free (Bolandra, Jepsonia) or ± adnate to ovary, usually not split to base (split to base in Tolmiea); sepals usually (4–)5(–6), distinct; petals usually (4–)5(–6) or absent, distinct, lobed or unlobed; nectary disc often encircling ovary distally at junction of ovary and free portion of hypanthium; stamens (2–)5(–9)10; anthers usually dehiscent longitudinally, rarely by broad terminal openings (Leptarrhena); staminodes absent; pistils 1, sometimes appearing 2–3+ (Micranthes), usually 2-carpellate, rarely 3-carpellate (Astilbe, Lithophragma, Micranthes), carpels connate for full length of ovary to barely connate proximally, equal, rarely unequal (Tiarella); ovary superior to inferior, 1–2(–3)-locular, ovaries fully connate when 1-locular, proximally connate to varying degrees when 2- and 3-locular (Astilbe, Micranthes); placentation axile, appearing marginal when ovaries are barely connate, or parietal; ovules anatropous, usually bitegmic, rarely unitegmic (most Micranthes), crassinucellate; styles 2–3(–4), distinct or connate (Saxifragopsis); stigmas 2–3(–4), capitate. Fruits capsular, sometimes folliclelike (Cascadia, Micranthes), 2–3(–4)-beaked, equally valvate (unequally valvate in Tiarella), dehiscence septicidal between beaks. Seeds 5–200, tan, brown, dark brown, black, yellowish brown, reddish brown, or red, rarely winged (Astilbe, Jepsonia, Sullivantia), ellipsoid, fusiform, ovoid, oblong, spheroid, oblong-cylindric, flat, or straight on 1 side, convex on other, rarely prismatic, smooth, wrinkled, ribbed, papillate, pitted, or ridged, tuberculate, warty, spiny, cellular-rugulose, or muricate; embryo straight; endosperm oily, copious.
Distribution
Nearly worldwide, primarily of north-temperate, arctic, and alpine regions.
Discussion
Genera ca. 38, species ca. 600 (23 genera, 158 species in the flora).
Classification of Saxifragaceae has been varied and controversial (e.g., A. Cronquist 1981; H. G. A. Engler 1930; J. Hutchinson 1973; G. K. W. Schulze-Menz 1964b; A. L. Takhtajan 1997; R. F. Thorne 1992). Molecular phylogenetic data (D. R. Morgan and D. E. Soltis 1993; Soltis et al. 1993, 2001; Angiosperm Phylogeny Group 1998, 2003) reveal that genera of Saxifragaceae in the broad sense are allied with at least ten separate, often distantly related families of flowering plants. These data also suggest that Saxifragaceae in the narrow sense as treated here consists of about 38 genera worldwide, equivalent to subfamily Saxifragoideae, one of the 15 subfamilies recognized by Engler and one of the 17 recognized by Schulze-Menz of the broadly defined Saxifragaceae. Molecular phylogenetic data (Soltis et al. 2001) show that the narrowly defined Saxifragaceae fall into two major groups: Saxifraga, and the heucheroid clade encompassing all other genera. Molecular data further show that Saxifraga, as traditionally understood, is polyphyletic, comprising two distinct lineages (treated here as Saxifraga and Micranthes) and the monospecific North American Cascadia. The major split between Saxifraga and the heucheroid clade is supported not only by molecular data from six DNA regions but by differences in patterns of floral morphology. Saxifraga has a relatively uniform floral morphology (radially symmetric flowers, with bilateral symmetry restricted to one Asian group of species, which consistently have the same number of sepals, petals, stamens, and carpels). Almost all of the variation in the family in numbers of sepals, petals, stamens, and carpels occurs in the heucheroid clade. Radially symmetric flowers predominate there, but some bilateral flowers are found in Bensoniella, Micranthes, Tolmiea, and some species of Heuchera.
Penthorum, the only genus in Penthoroideae of Saxifragaceae (H. G. A. Engler 1930), is morphologically anomalous in the Saxifragaceae and has often been included in Crassulaceae or, as treated here, its own family, Penthoraceae (Angiosperm Phylogeny Group 1998). Itea and Ribes, sole members of Iteoideae and Ribesoideae, respectively, are treated here as separate families, Iteaceae and Grossulariaceae. Crassulaceae, Grossulariaceae, Iteaceae, Paeoniaceae, Penthoraceae, and Saxifragaceae belong to the Saxifragales, as treated here, as well as Altingiaceae, Cercidiphyllaceae, Daphniphyllaceae, Haloragaceae (which includes Penthoraceae and Tetracarpaeaceae), Hamamelidaceae, Peridiscaceae, and Pterostemonaceae (Soltis et al. 2005).
Carpenteria, Decumaria, Deutzia, Fendlera, Fendlerella, Hydrangea, Jamesia, Philadelphus, and Whipplea once belonged to Hydrangeoideae of Saxifragaceae (H. G. A. Engler 1930) and are treated in the flora as Hydrangeaceae. Escallonia, the sole genus of Escallonioideae (Engler), is treated in the flora as Escalloniaceae. Molecular, morphological, and chemical data (C. R. Bensel and B. F. Palser 1975, 1975b, 1975c, 1975d; B. A. Bohm et al. 1988; M. L. Haskins and W. J. Hayden 1987; L. Hufford and W. C. Dickison 1992; D. R. Morgan and D. E. Soltis 1993; Soltis and B. A. Bohm 1982; Soltis et al. 1993; Soltis and P. S. Soltis 1997; Angiosperm Phylogeny Group 1998, 2003) indicate that Hydrangeaceae and Escalloniaceae are more likely to be related to the asterids, within which Hydrangeaceae appears to be close to Cornales.
Parnassia and Lepuropetalon have been included in Saxifragaceae as the subfamily Parnassioideae in the past (A. Cronquist 1981; H. G. A. Engler 1930; J. Hutchinson 1973; S. A. Spongberg 1972). Based on molecular, morphological, and chemical data, these genera appear to be only distantly related to other genera of Saxifragaceae and are here moved to Parnassiaceae (C. R. Bensel and B. F. Palser 1975, 1975b, 1975c, 1975d; B. A. Bohm et al. 1986; D. R. Morgan and D. E. Soltis 1993; Soltis et al. 2001; A. L. Takhtajan 1969; R. F. Thorne 1992). Molecular phylogenetic analyses (M. W. Chase et al. 1993; Zhang L. B. and M. P. Simmons 2006) have aligned Parnassiaceae with Celastraceae, either as a sister family or as a basal member of Celastraceae.
Molecular systematic studies have revealed that the approximately 38 remaining genera form a strongly supported clade (D. E. Soltis et al. 1993, 2000; Soltis and P. S. Soltis 1997; S. B. Hoot et al. 1999; V. Savolainen et al. 2000) that corresponds to the Saxifragoideae of Engler and is identical to the family circumscriptions of A. L. Takhtajan (1969, 1997) and R. F. Thorne (1992).
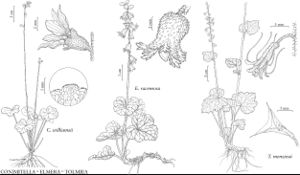
H. G. A. Engler’s (1930) subfamily Saxifragoideae contained one tribe, Saxifrageae, which consisted of four subtribes: Astilbinae, Leptarrheninae, Saxifraginae, and Vahliinae. Vahliinae is now known to be very distantly related to the Saxifragaceae. G. K. W. Schulze-Menz (1964b) elevated three of Engler’s subtribes to tribes: Astilbeae, Leptarrheneae, and Saxifrageae. K. Klopfer (1973) later recognized two large groups, one centered around Heuchera having parietal placentation, another centered around Saxifraga having axile placentation. Recent molecular phylogenetic and systematic studies indicate the presence of six well-marked clades, informally recognized as the Astilbe, Boykinia, Chrysosplenium, Darmera, Heuchera, and Leptarrhena groups (reviewed in Soltis et al. 2001). Relationships within some of these groups (e.g., the Boykinia and Heuchera groups) have also been studied in detail from a phylogenetic standpoint (e.g., Soltis and R. K. Kuzoff 1995; Soltis et al. 1997).
During fruit development in some genera (especially Saxifraga and Lithophragma), partially to mostly inferior ovaries swell to become increasingly superior due to allometric shifts (R. K. Kuzoff et al. 2001). Ovary position in keys and descriptions here refers to flowers during or shortly after anthesis. Reports of variation in ovary position in some genera and species may be due to observations of flowers at different developmental stages. D. E. Soltis et al. (2005) reported that most genera of Saxifragaceae display appendicular epigyny in floral development, which begins with a minute convex floral apex. During or after perianth initiation, the floral apex becomes concave, giving rise to an inferior ovary. Ovaries in Saxifragaceae often appear to be nearly superior or one-half to three-fourths inferior, but with their appendicular epigynous ground plan, they are not truly superior or homologous to superior ovaries. Instead, they are “superior mimics” with “pseudosuperior” ovaries.
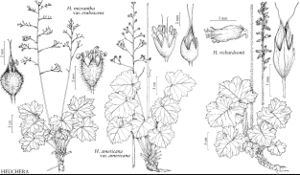
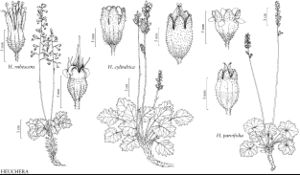
Species in some North American genera (particularly Boykinia, Darmera, Heuchera, Micranthes, Saxifraga, Tellima, Tiarella, and Tolmiea) are popular horticultural subjects.
Selected References
Lower Taxa
Illustrations
Key
| 1 | Leaves ternately decompound or 3-pinnately compound, never simple; flowers usually unisexual, sometimes bisexual; inflorescences plumose panicles, relatively large, branched, rarely congested and spikelike. | Astilbe |
| 1 | Leaves usually simple, rarely compound; flowers usually bisexual; inflorescences solitary flowers or racemes, panicles, simple or compound cymes, or thyrses | > 2 |
| 2 | Sepals 4; petals absent; stamens 2-8, usually 4 or 8; hypanthia greenish or yellow-green. | Chrysosplenium |
| 2 | Sepals 5(-6); petals (1-)4-5 or absent; stamens 3, 5(-6), or 10; hypanthia usually green, greenish, white, cream, yellow, yellowish green, pink to red or purple, or pinkish to reddish purple | > 3 |
| 3 | Stamens 3; petals 4; hypanthia ± split to base; adventitious buds usually produced at apices of petioles of basal rosette and cauline leaves, sometimes forming plantlets. | Tolmiea |
| 3 | Stamens usually 5 or 10; petals usually 5 or absent; hypanthia not split to base; adventitious buds and plantlets not present at apices of petioles of basal and cauline leaves | > 4 |
| 4 | Leaves peltate. | Darmera |
| 4 | Leaves not peltate | > 5 |
| 5 | Flowers heterostylous (stamens proximal to stigmas in some plants, distal in others); flowering in autumn(-winter) after basal leaves have withered, rarely flowers and leaves present together. | Jepsonia |
| 5 | Flowers homostylous; flowering in spring, summer, or autumn with leaves usually present | > 6 |
| 6 | Placentation parietal; ovaries 1-locular; inflorescences terminal from axillary buds in rosettes | > 7 |
| 6 | Placentation axile or marginal; ovaries 2-3-locular; inflorescences terminal from terminal buds in rosettes | > 15 |
| 7 | Stamens 10; inflorescences usually racemes or panicles, sometimes solitary flowers | > 8 |
| 7 | Stamens 5(-6); inflorescences racemes or thyrses, sometimes resembling panicles or spikes | > 11 |
| 8 | Styles 3; pistils 3-carpellate; sometimes with bulbils in axils of cauline leaves. | Lithophragma |
| 8 | Styles 2; pistils 2-carpellate; bulbils absent in axils of cauline leaves | > 9 |
| 9 | Capsules unequally valvate; petals unlobed; ovaries superior; leaves simple or 3-foliolate. | Tiarella |
| 9 | Capsules equally valvate; petals lobed; ovaries 1/4-1/2 inferior or nearly superior; leaves simple | > 10 |
| 10 | Petals 5-7-lobed; flowering stems 40-90 cm; hypanthia 4.5-9 mm; styles 1-1.5 mm; cauline leaves 2-3. | Tellima |
| 10 | Petals 9-11(-15)-lobed; flowering stems (2-)8-48 (-60) cm; hypanthia (0.6-)0.8-2.7 mm; styles 0.1-0.6 mm; cauline leaves absent or 1-3. | Mitella |
| 11 | Inflorescences thyrses (with cymose side branches), sometimes diffuse (resembling panicles) or dense (resembling spikes); petals unlobed, sometimes absent. | Heuchera |
| 11 | Inflorescences racemes; petals unlobed or lobed, rarely absent | > 12 |
| 12 | Hypanthia 5-9 mm; ovaries 1/4 inferior; petals 4-7-lobed, rarely ± unlobed; rocky ledges, cliffs, alpine open slopes, talus slopes; British Columbia, Oregon, Washington. | Elmera |
| 12 | Hypanthia 0.6-4 mm; ovaries 1/3 to nearly completely inferior; petals 3-11-lobed or unlobed, sometimes absent; moist sites and fir forests from Alaska, British Columbia, and Yukon to Alberta, California, Colorado, Oregon, South Dakota, and Utah, rocky slopes in the Rocky Mountains | > 13 |
| 13 | Styles 0.1-1.1 mm; petals usually 3-11-lobed, sometimes unlobed; hypanthia 0.6-2.7 mm; moist sites; Alaska to South Dakota, Utah, Colorado, California. | Mitella |
| 13 | Styles absent or 2-3 mm; petals usually unlobed, sometimes absent; hypanthia 2-4 mm; wet meadows, fir forests in California and Oregon, rocky slopes in the Rocky Mountains | > 14 |
| 14 | Styles 2-3 mm; petals linear, sometimes coiled, sometimes absent; hypanthia campanulate; capsules (1.8-)2-2.7 mm; ovaries 1/3 inferior; wet meadows, fir forests; California, Oregon. | Bensoniella |
| 14 | Styles absent; petals elliptic to narrowly spatulate; hypanthia cylindric to obconic; capsules 6.5-10 mm; ovaries 1/2-3/4 inferior; rocky slopes; Rocky Mountains from Alberta to Colorado. | Conimitella |
| 15 | Stamens 5 | > 16 |
| 15 | Stamens 10 | > 19 |
| 16 | Plants bearing bulbils at base of slender caudices, without rhizomes or stolons | > 17 |
| 16 | Plants not bearing bulbils at bases of caudices, rhizomatous and, sometimes, stoloniferous | > 18 |
| 17 | Sepals 3-10 mm; hypanthia 4-10 mm; capsules 8-11 mm; styles 1-2 mm; ovaries nearly superior; petals narrowly lanceolate, greenish with purple margins or reddish purple to dark purple. | Bolandra |
| 17 | Sepals 1.5-3.5 mm; hypanthia 2.5-3.5 mm; capsules 4-7 mm; styles absent or to 1 mm; ovaries 1/2-3/4 inferior; petals elliptic to obovate, white or pink, purple, or violet. | Suksdorfia |
| 18 | Seeds linear-fusiform, narrowly wing-margined, surfaces smooth; flowering stems and petioles glabrate or sparsely stipitate-glandular; cauline leaves glabrate; ovaries 1/2-4/5 inferior; petals 1.6-3.5 mm. | Sullivantia |
| 18 | Seeds ellipsoid, not winged, surfaces usually tuberculate, smooth in 1 species; flowering stems and petioles usually densely stipitate-glandular and eglandular-pubescent; cauline leaves stipitate-glandular; ovaries 2/3 to completely inferior; petals 2-12(-15) mm. | Boykinia |
| 19 | Anthers dehiscent by broad, terminal openings; carpels connate proximally; seeds fusiform, ribbed (at least over embryo). | Leptarrhena |
| 19 | Anthers dehiscent by longitudinal slits; carpels usually connate in proximal 1/4-1/2; seeds oblong, ovoid, or ellipsoid, smooth, ribbed, tuberculate, papillate, or spiny | > 20 |
| 20 | Hypanthia free from ovary 1-3.5 mm; stipules expanded from petiole bases; petals crimson-purple to violet-purple | Telesonix |
| 20 | Hypanthia completely adnate to ovary or free from ovary to 0.2 mm; stipules barely expanded or absent; petals white, cream, greenish, yellow, yellowish green, orange, red, pink, or purple, sometimes red-tinged or with purple margins or tips; sepals usually green, sometimes purple, sometimes reddish at tips | > 21 |
| 21 | Petioles jointed distally at attachment to leaf blades, separating early from blades; hairs unicellular; Siskiyou Mountains of California and Oregon, and sometimes in Washington. | Saxifragopsis |
| 21 | Petioles not jointed distally at attachment to leaf blades, not separating from blades; hairs multicellular, or plants glabrous; widespread, mostly north-temperate, arctic, and alpine regions | > 22 |
| 22 | Leaves cauline; flowering stems trailing or suberect, leafy; carpels connate proximally; seeds spiny; Coast Range to the western slopes of the Cascade Range from extreme nw California to sw Washington. | Cascadia |
| 22 | Leaves basal, or basal and cauline; flowering stems ± erect, leafless or leafy; carpels connate in proximal 1/2; seeds smooth, tuberculate, papillate, or ribbed; mostly north-temperate, arctic, and alpine regions | > 23 |
| 23 | Leaves usually basal; flowering stems usually leafless, rarely leaves crowded proximally at base of flowering stems; seeds ribbed. | Micranthes |
| 23 | Leaves basal and cauline; flowering stems usually with reduced cauline leaves; seeds smooth, tuberculate, or papillate. | Saxifraga |